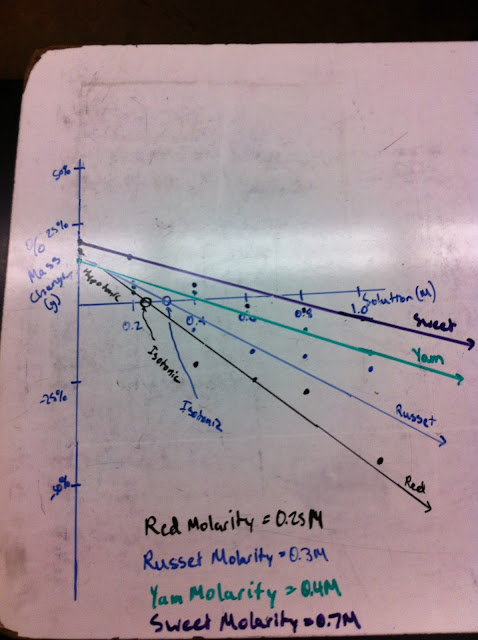
In class on Tuesday, we paired up into groups and did a lab. We had the option between two labs, both dealing with common ancestry. Brian and I chose to do the lab that involved the creation of DNA strands with the use of different colored beads and a pipe cleaner. We made 4 in total, one being human, one chimpanzee, one gorilla, and one the common ancestor. We found that the chimpanzee and the human were the most closely related, and that all four species shared some common bases, so they all shared the same common ancestor. We clarified that the more similar the genes and DNA of the species, the more similar they are alike in nearly all features.
 |
| In order from top down, here are small sections of the gene that codes for the hemoglobin proteins of Humans, Chimpanzee, Gorilla, and Common Ancestor |
Quiz Time
Part of our homework for tonight was to take a quiz on our blog,
so here goes my best attempt.
My Answers
1. The above picture shows morphological features shared between a common ancestor and its
descendants. There are homologous traits that can be found between all four of these species,
such as a similarity in arm structure. They all share the one bone, two bone, lots of bones
feature. When looking from the Mesonychid, to the Ambulocetus, to the Rodhocetus, to the
Basilosaurus, it can be seen that features such as the back legs did not appear and disappear
randomly, but rather gradually morphed over time, ending with the small flippers
at the base of the tail of the Basilosaurus.
2. e. North America
3. A dragonfly, a bird, and a bat all developed wings not as a result of homology, but rather
through convergent evolution. These shared traits are analogous, in that they formed as a
result of their environment, and did not originate from a common ancestor. Though they all
have wings, neither is quite similar to the other. A dragonfly has two wings on each side, a
bird has feathers on their wings, and a bat has a bone structure in their wings that is more
similar to that of a human arm. These three species gained their wings separately, as simple
adaptations to survive their environment. There is no direct link between any of them, they
did not evolve from each other.
4. In the Common Ancestor Lab, we created four different strands that each represented a small
section of the gene that codes for hemoglobin protein. Each hemoglobin strand represented a
different animal. We made one strand for a human, one for a chimpanzee, one for a gorilla,
and one for their common ancestor. The human and chimpanzee strands were the closest in
comparison, which explains why they appear to be very similar in structure, intelligence, and
more. With the common ancestor having the least similar DNA to humans, the
chimpanzee having the closest, and the gorilla somewhere in the middle, it can be concluded
that evolution exists, and that humans evolved from their common ancestor. While some of
the DNA experiences no change, other parts of the DNA change, leading to the formation of
a new species. This new formed species then experiences changes in its own DNA, and
forms yet another new species. DNA proves evolution, because similar DNA structures must
have originated from somewhere, and that somewhere is the common ancestor.
5. Homology is the study of the structural similarities or gene similarities between two or more
species, caused as the result of sharing a common ancestor. These similar features, otherwise
known as homologous features, are similar because they are inherited from a common
ancestor. These homologous features simply morph from this ancestor, they do not become
something entirely different. For example, the hand of a human and the hand of an ape are
said to be homologous, for they both inherited the characteristics of the hand of their common
ancestor. They cannot just change into wings over night. It would take millions and millions
of years for something like that to occur. In Your Inner Fish, an excellent example that shows
homology would be the one bone, then two bones, then blob of bones, then fingers structure
of the arm, as discovered by Sir Richard Owen. This structure can be seen in humans, seals,
lizards, birds, bats, penguins, and whales, among many other species. These similarities in
structure are no coincidence, but rather they all originated from a common ancestor with this
trait. It seems that this common ancestor was Tiktaalik, for it served as the transition species
from water to land, and was the first known species to have an arm such as this. Essentially,
all of the above species developed there arms similar to that of Tiktaalik, and therefore they
all share this homologous trait.
For homework, we also had to read Survival of the Sickest, chapter 4, and come up with a way to summarize it. I decided to do a poem. I added this poem below.
The buzzing sound of a million deaths,
was stopped at the site of a bean!
With the lack of an enzyme, the loss of red blood cells,
the blood would become too lean!
The tiny little beast, and G6PD deficiency,
two terrible diseases, many lives they have stole!
But together it’s true, throughout human evolution,
that this mutation has saved many souls!
Favism is so prevalent, and Pythagoras was right,
Eating fava beans was making his students sluggish!
Primaquine was the key, to the discovery of this disease,
in the Korean War where many soldiers perished!
But little did we know, that survival of the sickest
would lead to convergent evolution!
Favism would prevail, protection from malaria,
It was an illness that became
part of the solution!